|
|
| Electron Emission |
|
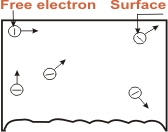
The liberation of electrons from the surface of a metal is
known as Electron Emission. If a piece of metal is
investigated at room temperature, the random motion of the
electrons will be shown in Fig. However, these electrons are
free to the extent that they may transfer from one atom to
another within the metal but they cannot leave the metal surface
to provide electron mission. It is because the free electrons
that start at the surface of metal find behind them positive
nuclei pulling them back and none pulling forward. Thus at
the surface of the metal , a free electron encounters forces
that prevent it to leave the metal. In other words, the metallic
surface offer a barrier to free electrons, their kinetic energy
increases and is known as surface barrier.
|
|
|
However, if sufficient energy is given to the free electrons,
their
kinetic energy increases and thus the electrons will cross over
the surface barrier to leave the metal. |
|
 |
|
|
Work function (W0): The minimum energy
required by an electron to just escape (i.e. with zero velocity)
from metal's surface is called Work function (W0)
of the metal. The work function of pure metals varies (roughly)
from 2eV to 6eV. Its value depends upon the nature of the metal,
its purity and the conditions of the surface. We selected those
metals for electron emission which have low work function. |
|
|
Types of Electron Emission: |
|
|
The electron emission from
the surface of a metal is possible only if sufficient addition
energy (equal to work function of the sources such as heat
energy, energy stored in electric field, light energy or kinetic
energy of the electric charges bombarding the metal surface.
Accordingly; there are following four principal method of
obtaining electron emission from the surface of a metal:
|
|
(I) Thermionic emission: In this method, the metal
is heated to a sufficient temperature (about 2500oC)
to enable the free electrons to leave the metal surface. The
number of electrons emitted depends upon the temperature.
The higher the temperature, the greater is the emission of
electrons. This type of emission is employed in vacuum tubes. |
|
(II) Field emission: In this method, a strong electric
field (i.e. a high positive voltage) is applied at the metal
surface which pulls the free electrons out of the metal because
of the attraction of positive field. The strong the electric
field, the greater is the electron emission. |
|
(III) Photoelectric emission: In this method, the energy
of light falling upon the metal surface is transferred to
the free electrons within the metal to enable them to leave
the surface. The greater the intensity of light beam falling
on the metal surface, the greater is the photoelectric
emission. Photoelectric
emission is utilized in photo tubes which from the basis of
television and sound films. |
|
(IV) Secondary emission: In this method, a high velocity
beam of electrons or other out. The intensity of secondary
emission depends upon the emitter material, mass and energy
of bombarding particles. |
|